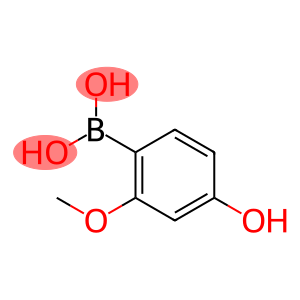
Boronic acid, B-(4-hydroxy-2-methoxyphenyl)-
4-hydroxy-2-methoxyphenylboronic acid
CAS: 550373-98-3
Molecular Formula: C7H9BO4
Boronic acid, B-(4-hydroxy-2-methoxyphenyl)- - Names and Identifiers
Boronic acid, B-(4-hydroxy-2-methoxyphenyl)- - Physico-chemical Properties
| Molecular Formula | C7H9BO4 |
| Molar Mass | 167.96 |
Boronic acid, B-(4-hydroxy-2-methoxyphenyl)- - Introduction
Acid is an organic compound with the chemical formula C7H9BO4.
Its properties are as follows:
-Appearance: White crystalline solid
-Melting point: 235-240°C
-Solubility: Soluble in water and organic solvents (such as ethanol, dimethyl sulfoxide, etc.)
- pH: stable under acidic conditions, unstable under alkaline conditions
Acid is often used as an important organic synthesis reagent in organic synthesis, mainly used in the following fields:
-Catalyst: It can be used to catalyze the conversion reaction between alcohol and aldehyde, such as Suzuki-Miyaura coupling reaction, Heck reaction, cycloaddition reaction, etc.
-Drug synthesis: It is widely used to prepare a series of pharmaceutically active molecules, such as anti-tumor drugs, anti-inflammatory drugs, etc.
Preparation Method:
there are many methods for preparing acid. one of the commonly used methods is to oxidize toluene to obtain p-methylhydroxytoluene, and then to obtain the target product through boration reaction.
Safety Information:
Because it is an organic compound, some safety measures need to be paid attention to when using and storing:
-Avoid contact with skin and eyes, wear appropriate protective gloves and glasses when using.
-Avoid inhaling its dust or gas and ensure that it is operated in a well-ventilated place.
-Keep sealed when stored, away from fire and oxidant.
-Comply with local safety and environmental regulations when using and disposing of waste.
Its properties are as follows:
-Appearance: White crystalline solid
-Melting point: 235-240°C
-Solubility: Soluble in water and organic solvents (such as ethanol, dimethyl sulfoxide, etc.)
- pH: stable under acidic conditions, unstable under alkaline conditions
Acid is often used as an important organic synthesis reagent in organic synthesis, mainly used in the following fields:
-Catalyst: It can be used to catalyze the conversion reaction between alcohol and aldehyde, such as Suzuki-Miyaura coupling reaction, Heck reaction, cycloaddition reaction, etc.
-Drug synthesis: It is widely used to prepare a series of pharmaceutically active molecules, such as anti-tumor drugs, anti-inflammatory drugs, etc.
Preparation Method:
there are many methods for preparing acid. one of the commonly used methods is to oxidize toluene to obtain p-methylhydroxytoluene, and then to obtain the target product through boration reaction.
Safety Information:
Because it is an organic compound, some safety measures need to be paid attention to when using and storing:
-Avoid contact with skin and eyes, wear appropriate protective gloves and glasses when using.
-Avoid inhaling its dust or gas and ensure that it is operated in a well-ventilated place.
-Keep sealed when stored, away from fire and oxidant.
-Comply with local safety and environmental regulations when using and disposing of waste.
Last Update:2024-04-09 21:04:16
Supplier List
Product Name: (4-Hydroxy-2-Methoxyphenyl)boronic acid Request for quotation
CAS: 550373-98-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 550373-98-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: (4-Hydroxy-2-Methoxyphenyl)boronic acid Visit Supplier Webpage Request for quotation
CAS: 550373-98-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 550373-98-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: (4-Hydroxy-2-Methoxyphenyl)boronic acid Request for quotation
CAS: 550373-98-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 550373-98-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: (4-Hydroxy-2-Methoxyphenyl)boronic acid Visit Supplier Webpage Request for quotation
CAS: 550373-98-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 550373-98-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

